News follows company’s clearance for 24-minute VH202 BI received in July
Goal is to enhance patient safety by making it easy to monitor every load
3M announced today its submission for 510(k) clearance with the U.S. Food and Drug Administration (FDA) for its Attest™ Super Rapid System for Steam Sterilization, which will provide biological indicator (BI) results in 24 minutes. The clearance of a faster readout time for steam will continue to enhance patient safety while increasing workflow advantages for sterilization professionals.
“The focus of our sterilization science at 3M is on increasing patient safety through every means possible,” began Srini Raman, 3M Business Director of Device Reprocessing. “One of the best ways we can do that is to make it easy for sterilization departments to know that every load of medical instruments has met standards for sterilization beforethey’re released for surgery. We introduced a 24-minute readout for VH202 in July and it was met with great market excitement. Our next goal was to take this same science and apply it to steam sterilization monitoring, which 3M has been a progressive leader in for over 40 years.”
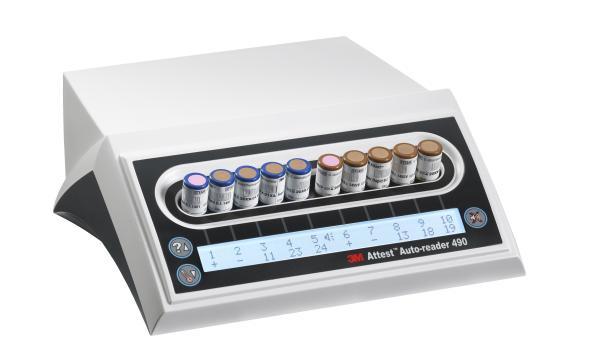
The addition of the 3M™ Attest™ 490 Auto-reader to 3M’s sterilization portfolio, pending 510(k) clearance, will mean customers can count on 3M for a simplified, standardized approach to BI processing for both Steam and VH202 results in just 24 minutes.
The new system will utilize existing hardware and will continue to use the same BI technology that customers use today.
3M and Attest are trademarks of 3M Company.
About 3M
At 3M, we apply science in collaborative ways to improve lives daily. With $32 billion in sales, our 90,000 employees connect with customers all around the world. Learn more about 3M’s creative solutions to the world’s problems at http://www.3m.com/ or on Twitter @3M or @3MNewsroom.
Multimedia Files: